  
The more randomness in a system means the entropy of the system is higher. A decrease in the thermal energy of these particular systems, that is, a fall in temperature. And entropy of liquid is higher than solid. This occurs through the Brownian motion of a nanoresonator. Its an extensive property of a thermodynamic system The following points must be considered when ranking the systems according to their entropy: The entropy of gases are highest than liquids or solid. The entropy change is unknown (but likely not zero), because there are equal numbers of molecules on both sides of the equation, and all are gases. Entropy can understood as the measure of randomness or disorder in a system. Explanation: Entropy is the measure of disorder or randomness in a closed system. If ∆S is negative, then the negative signs (from the subtraction and the sign of ∆S) will cancel out, and so as T∆S gets bigger, ∆G will get more positive.\left( g \right)\) T is always positive, so if ∆S is positive then a bigger T∆S will make ∆G more negative (since we subtract T∆S). As T increases, the T∆S component gets bigger. The energy of the system is spreading it has high entropy, whereas if the energy of the system remains concentrated it is said to have decreased entropy. In which of these systems is the entropy decreasing A. Explanation: Entropy refers to phenomenon which measures the spreading of energy in a system. The total entropy can remain constant in. The second law of thermodynamics states that the total entropy can only increase over time for an isolated system, meaning a system which neither energy nor matter can enter or leave. ∆H is still positive and ∆S is still whatever sign you figured out above. f Get an answer Search for an answer or ask Weegy. The entropy of the universe must always increase for an spontaneous reaction. Since ∆H and ∆S don't change significantly with temperature (given in the question), we can assume that they keep the same signs and values: i.e. 
It is one of the foundational concepts of chemistry and it is one of the fundamental laws of our universe. If ∆G is negative (from the question), is the reaction spontaneous or non-spontaneous?Ģ) Let's use ∆G = ∆H - T∆S again. This is known as the Second Law of Thermodynamics. a gas condensing to a liquid A gas condensing to a liquid: in this system, the entropy is decreasing. Collisions of these systems with land causes the orderly streaming gas to disperse. From these values, we can know for certain whether ∆S is positive or negative (hint: remember that we are subtracting ∆G!).ġ) Knowing the sign of ∆G is enough to say whether the reaction is spontaneous or not under these conditions. In which of these systems is the entropy decreasing A. In which of these systems is the entropy decreasing air escaping from a tire c. Gas capture such as this represents a huge decrease in entropy. Temperature is always positive (in Kelvin). 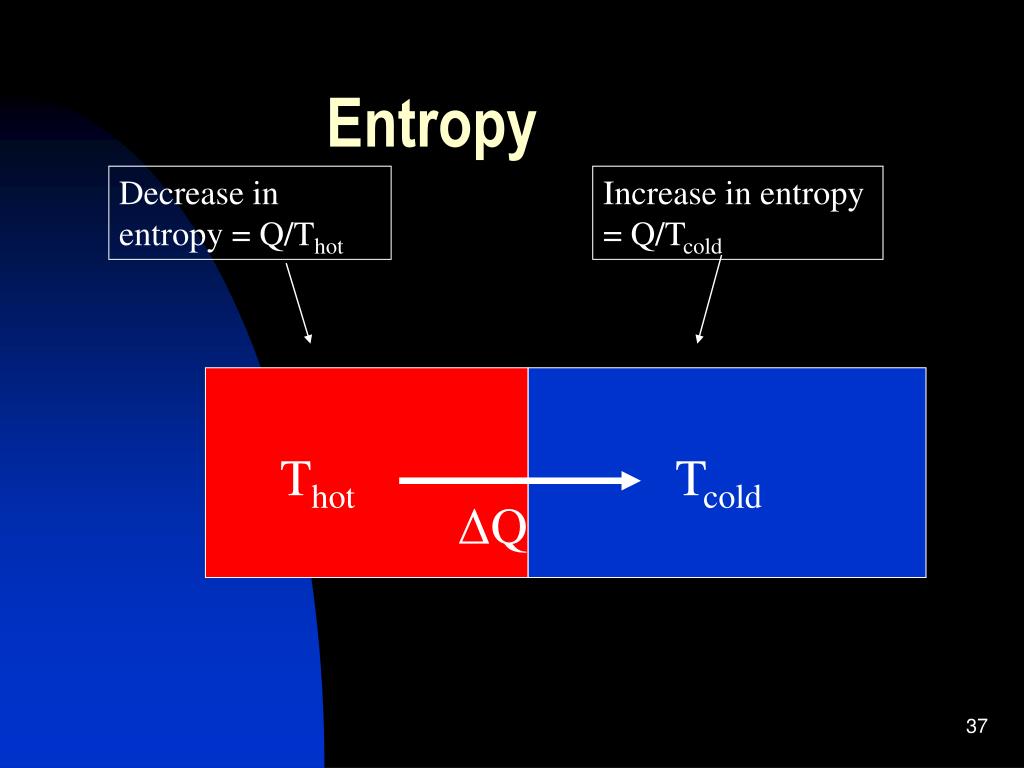
As the temperature increases, the entropy of the atoms in the lattice increase. The position of the atoms or molecules in the crystal would be perfectly defined. size of air is decreased without removing heat from the air (while Enthalpy remains same), this decrease in Entropy without decreasing Enthalpy i.e. We know (from the question) that ∆G is negative and that ∆H is positive. The entropy of a pure crystalline substance at absolute zero (i.e. In this process Entropy is decreased i.e. To rank items as equivalent overlap them. This looks like a homework question, so I'll give you some hints to get you on the riht path rather than answering directly.ģ) We know that ∆G = ∆H - T∆S. Rank these systems in order of decreasing entropy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
