  
The main difference between molecular geometry and electron pair geometry is that molecular geometry does not include unpaired electrons, whereas electron pair geometry includes both bonded atoms and unpaired electrons. Electron Pair Geometry: This is the 3-D arrangement of electron pairs around the central atom of a polyatomic ion or molecule.Molecular Geometry: This is the 3-D arrangement of bonded atoms in a polyatomic ion or molecule.Lone Pair: This refers to a pair of valence electrons that are not shared with another atom.Bond Angle: This is the angle between a bonded atom, the central atom, and another bonded atom.The following terms are commonly used in discussing the shapes of molecules. Each shape has a name and an idealized bond angle associated with it. 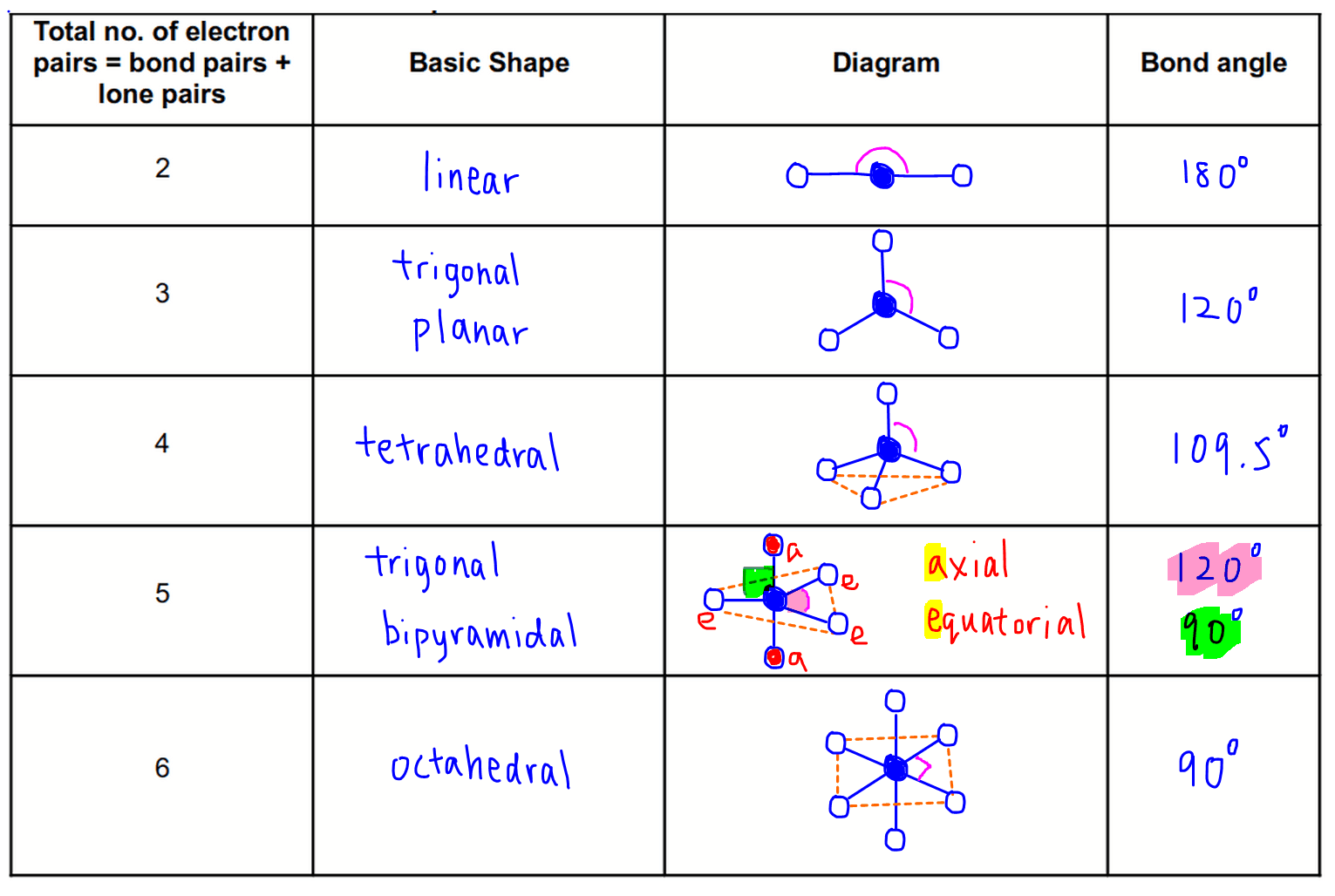
It can predict the shape of nearly all compounds that have a central atom, as long as the central atom is not a metal.VSEPR models are based on the concept that electrons around a central atom will configure themselves to minimize repulsion, and that dictates the geometry of the molecule.The VSEPR model predicts the 3-D shape of molecules and ions but is ineffective in providing any specific information regarding the bond length or the bond itself. Lewis structures only tell the number and types of bonds between atoms, as they are limited to two dimensions.Browse more Topics under Chemical Bonding And Molecular Structure It is useful for nearly all compounds that have a central atom that is not a metal. Specifically, VSEPR models look at the bonding and molecular geometry of organic molecules and polyatomic ions. It is basically a model to predict the geometry of molecules. The Valence Shell Electron Pair Repulsion Model is often abbreviated as VSEPR (pronounced “vesper”). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
